Why Climate Scientists were Duped into Believing Rising CO2 will Harm Coral and Mollusks
The pile of wrong facts, regarding Climate, is mountainous.
CORAL REEFSOCEAN ACIDIFICATION
Why Climate Scientists were Duped into Believing Rising CO2 will Harm Coral and Mollusks
Jim Steele
Coral build reefs by producing limestone, or calcium carbonate. The great diversity of shell building mollusks, like clams and oysters, also build their shells out of calcium carbonate. So, scientists assumed that these organisms just pulled carbonate ions from the surrounding sea water and joined it with abundant calcium ions to make reefs and shells, a process referred to as “calcification”. Thus, many scientists then expressed their heart-felt concerns that more CO2 will reduce the ocean’s carbonate ions and thus stress coral reef building and mollusk shell building.
Indeed, the increasing absorption of human produced carbon dioxide by the oceans can very slightly lower pH. In other words, more CO2 increases the oceans’ concentration of H+ ions. It is also unassailable science that when CO2 enters the water, it interacts with water molecules to produce both H+ ions and bicarbonate ions. However, those H+ ions can then interact with carbonate ions and convert them to also form bicarbonate ions and reduce the pool of available carbonate ions. So, NOAA and hundreds of internet websites falsely told the world that “Ocean acidification slows the rate at which coral reefs generate calcium carbonate, thus slowing the growth of coral skeletons.”
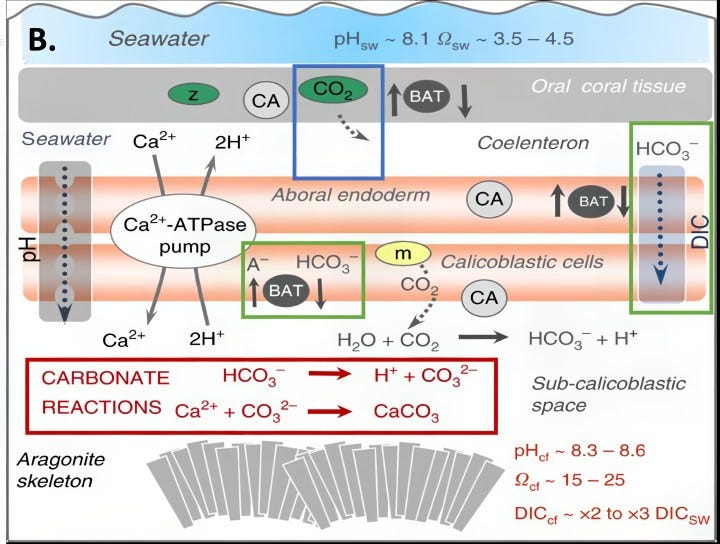
However, climate scientists were apparently very ignorant regards the physiology of reef building and shell making. In order for charged ions to pass through an organism’s lipid membranes and enter its calcification chambers, a specialized channel or transporter is required. But for over a decade now, the search for carbonate transporters has failed to find any such transporters in any of these organisms. However, abundant bicarbonate transporters (green rectangles) have been found and deemed important for making reefs and shells.
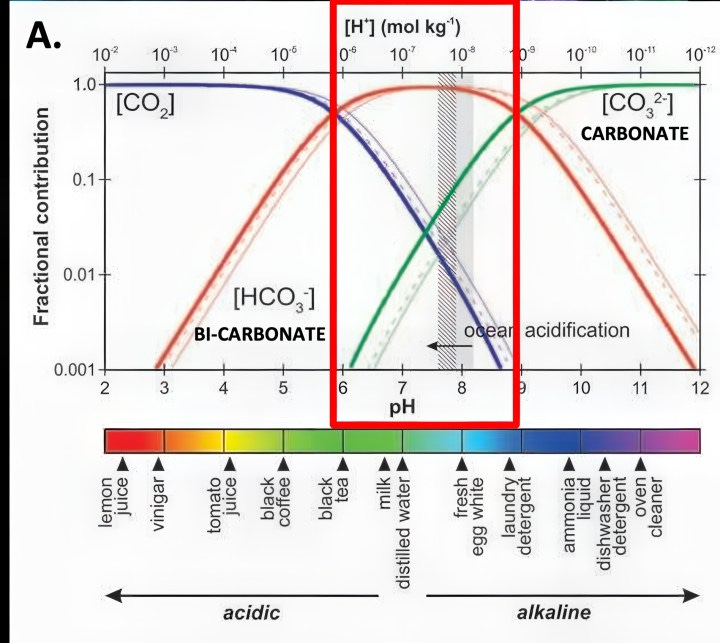
From an evolutionary perspective, using the more abundant bi-carbonate ions is extremely logical. The higher amounts of carbon dioxide in the ancient atmosphere would lower ocean pH when mollusks and coral first evolved millions of years ago. That argues using scarcer carbonate ions would be very risky but using much more abundant bi-carbonate ions would supply the stability to evolve. Furthermore, all those organisms had long had the ability to absorb bicarbonate ions and transform it internally into carbonate ions for shell making and reef building by simply pumping out hydrogen ions. When CO2 enters the oceans, over 90% of it converts to bicarbonate ions whether the oceans’ pH is acidic at pH 6 or basic at pH 9. In contrast, carbonate ions virtually do not exist when ocean pH approaches pH 6. (Graphic A.)
The world can only hope that NOAA and all those alarmist websites will soon admit that improved science has revealed the error of their ways, and they will now come clean and tell the public that CO2 has not threatened reef builders and shell builders. In fact, they should report that more CO2 generates more bicarbonate ions which are the building blocks of shells and reefs.




Great article!
The ocean covers 75 % of the planet and is a massive sink for CO2:
The CO2 carbonate cycle also features dropping out giga tons of insoluble carbonates to the bottom of the sea - a massive sink for CO2!
https://tucoschild.substack.com/p/update-coral-reef-recovery-disappoints
Geologists understand that the Devonian was one of the most prolific carbonate building periods in the last 600 million years with CO2 levels of 4000 ppm, followed closely by the Cretaceous with CO2 levels at 2500 ppm. This helps explain why reef building is more prevalent in the geologic record when CO2 levels are high.